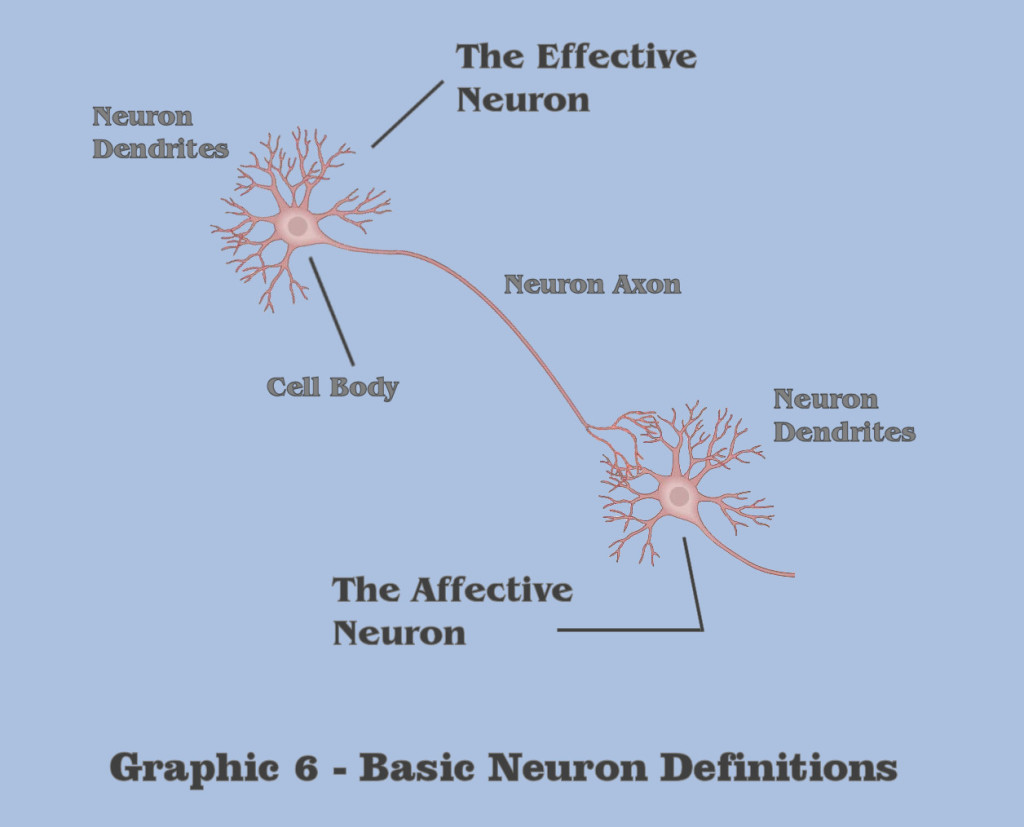
In graphic 6, we introduce some basic terminology that will be used throughout the visualization. And although all of the graphics in this dialog depict static expositions, mostly for purposes of defining terms, the conceptualizations that this dialog intends to convey are dynamic, and the interrelations underlying these dynamics are the true message.
Throughout this visualization, the discussion will make a primary discrimination which is somewhat unlike any of the designations used in traditional neuroscience, because the dialog wants to make to make a distinction between effective neuron behavior and the behavior in what is termed affective neurons. Although the designation between effective and affective is purely relative, the term ‘effective neuron’ will refer to the collective neural mechanics between a cell body and its axon, and the term ‘affective neuron’ will refer to the collective neural mechanics between a neuron cell body and its dendrites. For those students of massively asynchronous assemblies, these distinctions are fundamental to the understanding of abstraction in general, and critical to the induction of gestalt abstraction, and all of the conceptualizations discussed in this visualization shall apply in totality to the artificial assemblies we will be building for our artificial agent.
As a note: Although this chapter is focusing on the afferent, dendritic centered functionality of neural asynchrony, the enormous conversation on the phylogenetics and ontogeny of the complex dispersal efferents (the neural axons and their targeting of affective dendrites), which has historically had a large proportion of interest throughout much of neuroscientific research, is not discussed until later in the dialog, when the discussion begins the actual engineering of artificial intelligence.
In its most simplistic terms, a neuron in general has a singular output mechanism, called the axon, which signals a condition that is produced by the neurons’ input mechanism, its dendrites, a condition which we shall call the dendritic threshold. The dendritic threshold is considered to be a certain level of electrotonic charge which accumulates in the dendrite of neurons. Although it implies a charge magnitude at the dendrites themselves, in reality it is a measurement of accumulated charge at the cell body, or more specifically, what is referred to as the axon hillock, but for purposes of this depiction, the discussion will simplify the definition to be the summed charge of the cell body as accumulated by the neuron dendrite. Following this simplified characterization, this dialog is making the distinction in terms between the effective neural mechanics in axons and affective neural mechanics of dendrites due to a different set of molecular responses that Nature has chosen in creating effective charge potentials and affective charge potentials. Because Nature rarely chooses indiscriminately, the dialog wants the bottom-up engineer to keep this dichotomy in mind as the visualization proceeds.
The set of molecular responses that we are talking about at the neural level is, of course, the distribution of extracellular and intracellular ions that create charge formations, and it is curious that Nature has chosen different groups of ions to implement active charge formation in the axons of neurons than the groups it has chosen for charge formation in the dendrites of neurons. Specifically, the dialog has already mentioned the active mechanisms that utilize sodium and potassium ions for the propagation of charges throughout the axons of neurons, and it is significant that Nature has chosen a different mix of ions for its active charge formation in the dendrites of neurons. Specifically, calcium ions are added to the mix of sodium and potassium ions (and chlorine ions in the case of inhibitive neural signaling, but that mechanism will not be a subject in this current discussion) in the dendritic response to charge formation.
Because of their differentiation, the Organon Sutra will maintain a distinction between effective axonic behaviors and affective dendritic behaviors in the dialog. And in light of that distinction, the visualization will proceed first with a focus on the dendrites and the afferent mechanisms of basic neural asynchrony.
The dendrites in affective neurons trigger the dendritic threshold by accumulating individual charge depolarizations that occur from a number of effective neuron axons that synapse to a dendrite. In this visualization, the dialog will initially define a neurons’ dendrites as having an isomorphic response characteristic, in that they respond equally to any synapse, because we will want to initially separate the dendritic summation properties from the state and state retention properties which we are currently seeking to define.
Now, before we can talk of a charge accumulation, or this accumulation of input signals, the dialog must first define what is meant by an individual depolarization. It is here that we will want to find the neurological inflection point where the temporally dimensionless axonic impulse is given dimension by the numerous mechanisms of the dendrite, the first of which we shall call the ionotropic time course of individual depolarizations.
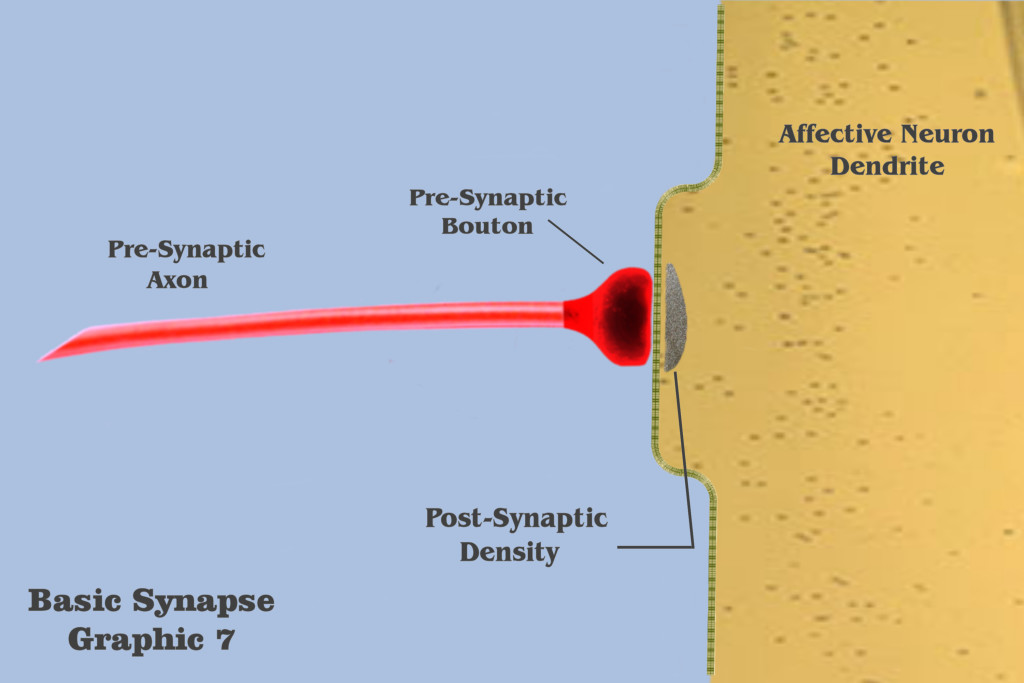
Graphic 7 presents some terms and depicts those elements to be used in the visualization of an individual ionotropic charge depolarization.
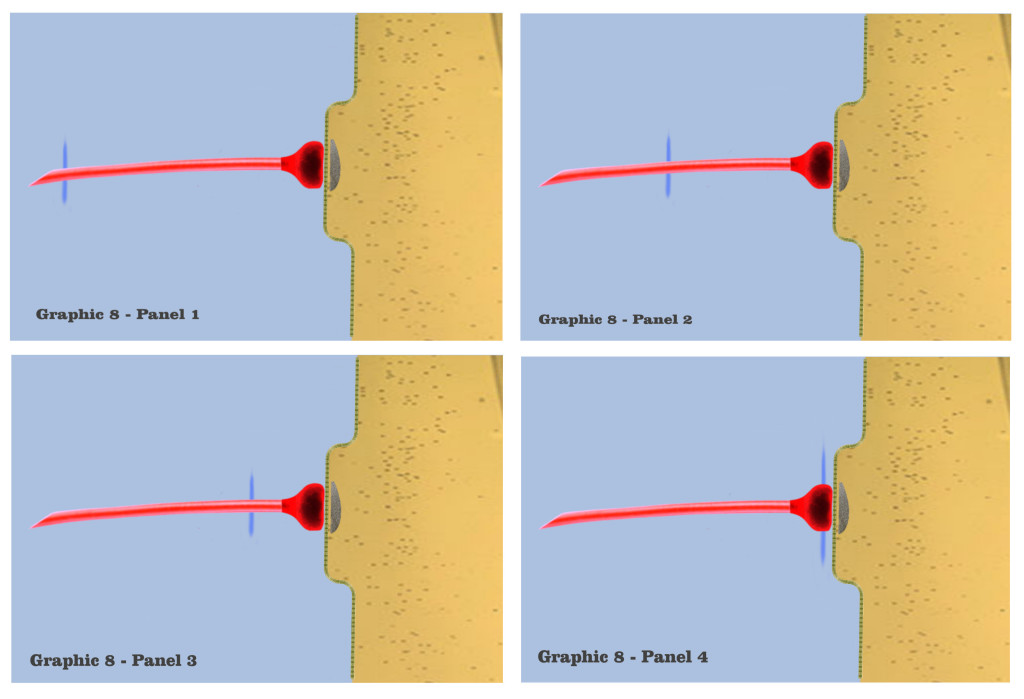
And in panel 1 of graphic 8, the dialog begins the visualizations of the ionotropic time course with a depiction of the axonic impulse as it is approaching the isolated dendritic synapse. The electrogenic propagation of the impulse is represented by a bluish wave emanating from the presynaptic axon. Although the colorization itself is entirely arbitrary, the bluish hue has been chosen to represent the efferent charge potentials of axons throughout the visualization, and to discriminate effective charge potentials from affective charge potentials.
In the second panel of graphic 8, we see this axonic impulse as it is traveling further along the axonic conduit, and in panel 3 it approaches the presynaptic bouton. In panel 4, the axonic impulse has arrived at the presynaptic bouton, to set in motion the mechanisms that will effect the time course of an ionotropic depolarization. Many of these mechanisms have already been introduced in the dialog, such as vesicle release during exocytosis, and the intricate process of neurotransmitter re-uptake into the bouton during endocytosis, but the only presynaptic mechanism to have bearing on the visualization is the release of vesicles from the bouton to the postsynaptic density.
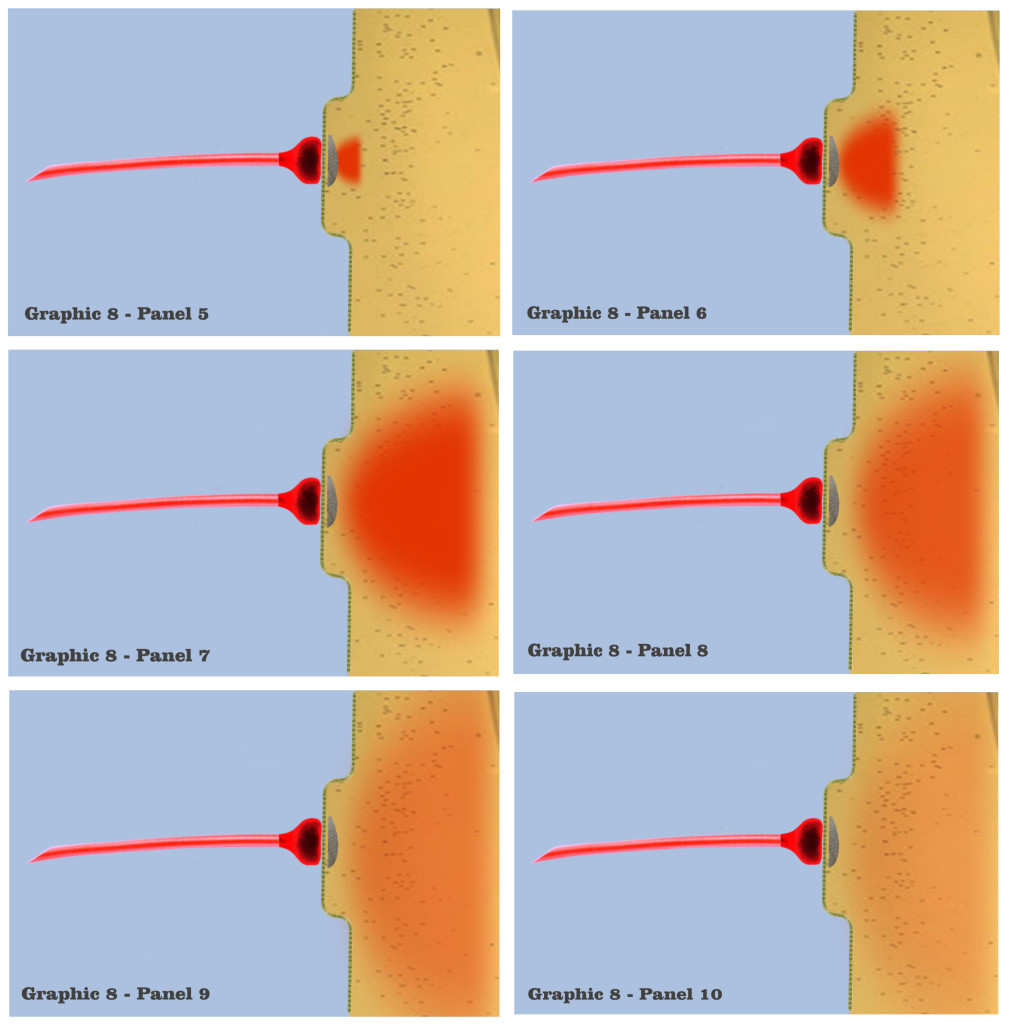
Although the graphic in panel 5 does not depict the vesicles themselves, or their subsequent saturation of the postsynaptic site, panel 5 shows the resultant charge potential which is forming from ionotropic receptors in the postsynaptic membrane, as they respond to the presynaptic release of neurotransmitter. Charge formation in the postsynaptic dendrite is accomplished by a depolarizing concentration of sodium, potassium and calcium ions which enter the dendritic membrane through ionotropic receptors, the concentrations being represented by the red-shaded field in the graphic. The red hue in the graphic, although again arbitrarily chosen, represents the charge fields of this greater mix of ions, just as the blue hue has been chosen to represent axonic charge potentials.
In panel 6, we see the depolarization concentration growing, as the graphic depicts the first phase in the time course of an individual synaptic depolarization. The concentrations of ions that flow into the postsynaptic site vary according to the variety of ionotropic receptors at a particular synapse, but since this visualization has characterized the depicted dendrites as functionally isomorphic, variations of initial concentrations do not bear on the scenario. Regardless of the type of ionotropic receptor, it is the time course of the postsynaptic charge dissipation that is being considered in the visualization.
In panel 7, we visualize the complete induction of charge ions that have been admitted past the dendritic membrane, to spread into a maximal charge field, which marks the second phase of the time course in individual charge depolarizations. The beginning of this second phase is depicted in panel 8, where the maximal charge field begins to spread, losing intensity as it spreads. Now, although it should be noted that the electro-physics of true electrotonic spread occurs predominantly along the capacitive dendritic membrane, the graphics will retain a visualization of charge spread as occurring in a roughly homogenous field.
And in panel 9, we see a depiction of the electrotonic charge dissipating somewhat, as the charge potential spreads along the membrane and its intensity diminishes. This charge dissipation continues in panel 10, after which there would be a complete dissipation of the charge ions as they diffuse throughout the dendritic medium.
It is a visualization of this depicted time course for an individual charge depolarization that the bottom-up engineer should keep in mind, as it represents the basic process which occurs when an axonic action potential triggers a postsynaptic depolarization in the absence of any other spatially or temporally grouped action potentials at that dendrite. Although viewed in isolation, the bottom-up engineer should also know that the complete dissipation of an individual depolarization would not provide enough charge potential for the neuron as a whole to reach its dendritic threshold. The visualization provides a frame work to illustrate how collective depolarizations do trigger a dendritic threshold, and can subsequently illustrate the mechanisms for synaptic persistence that we are building to.
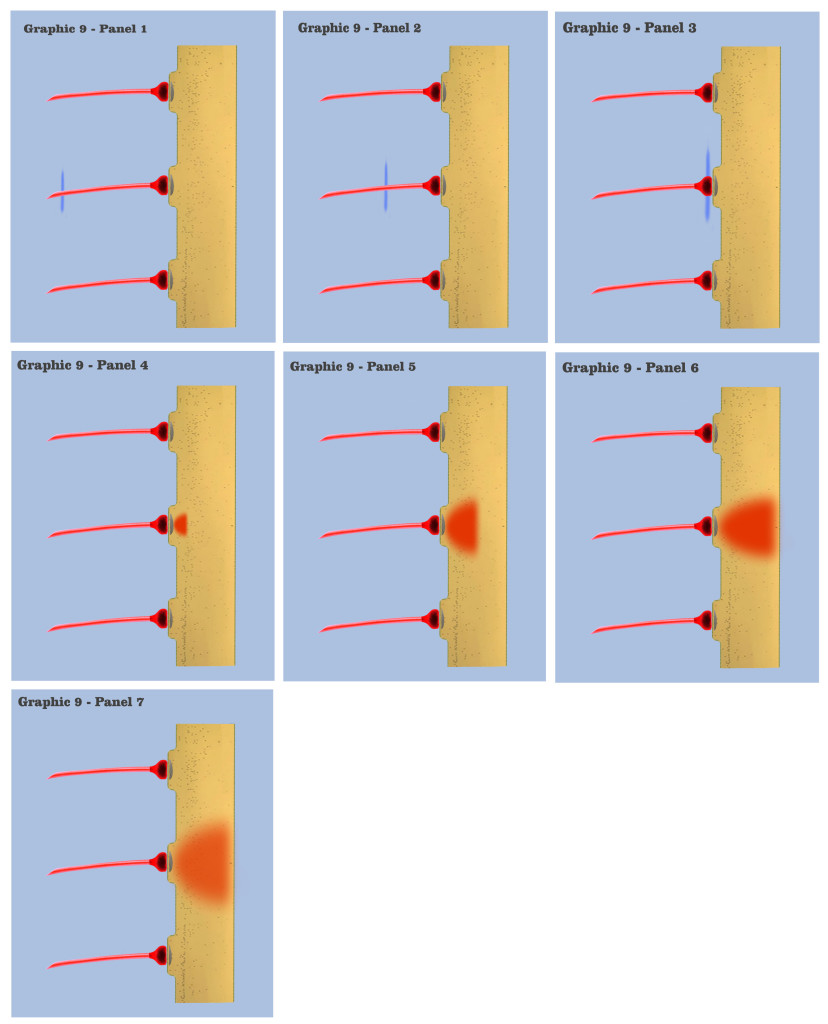
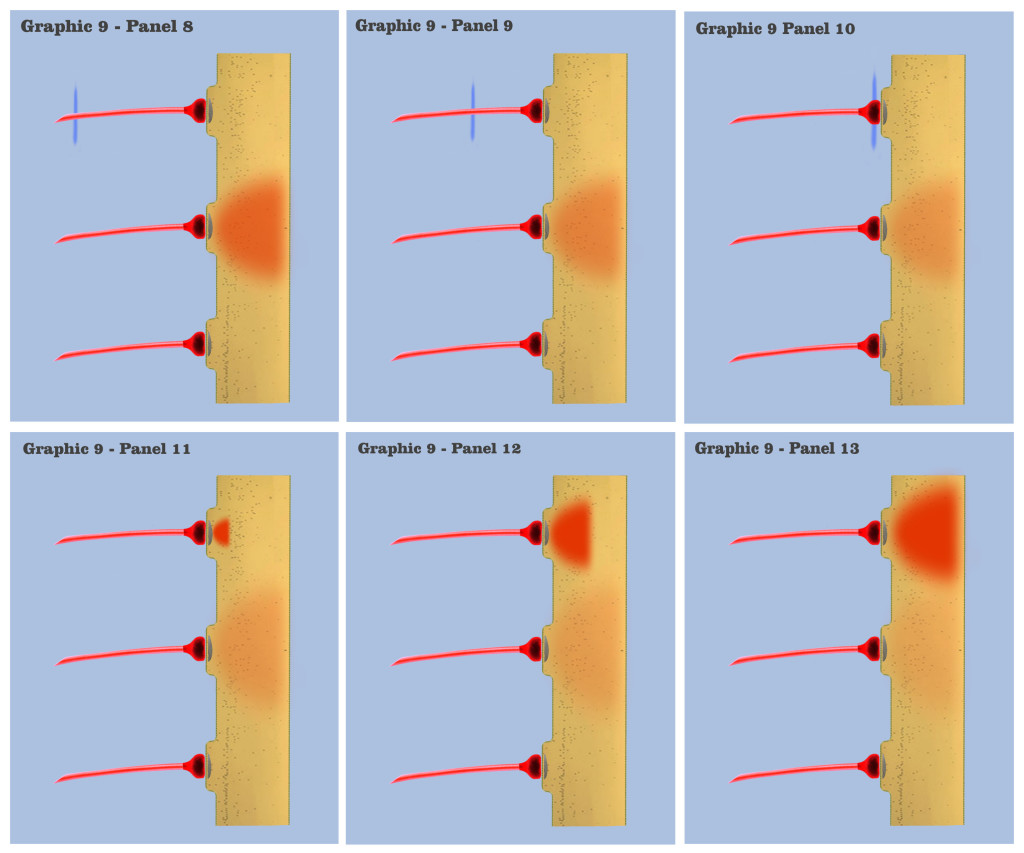
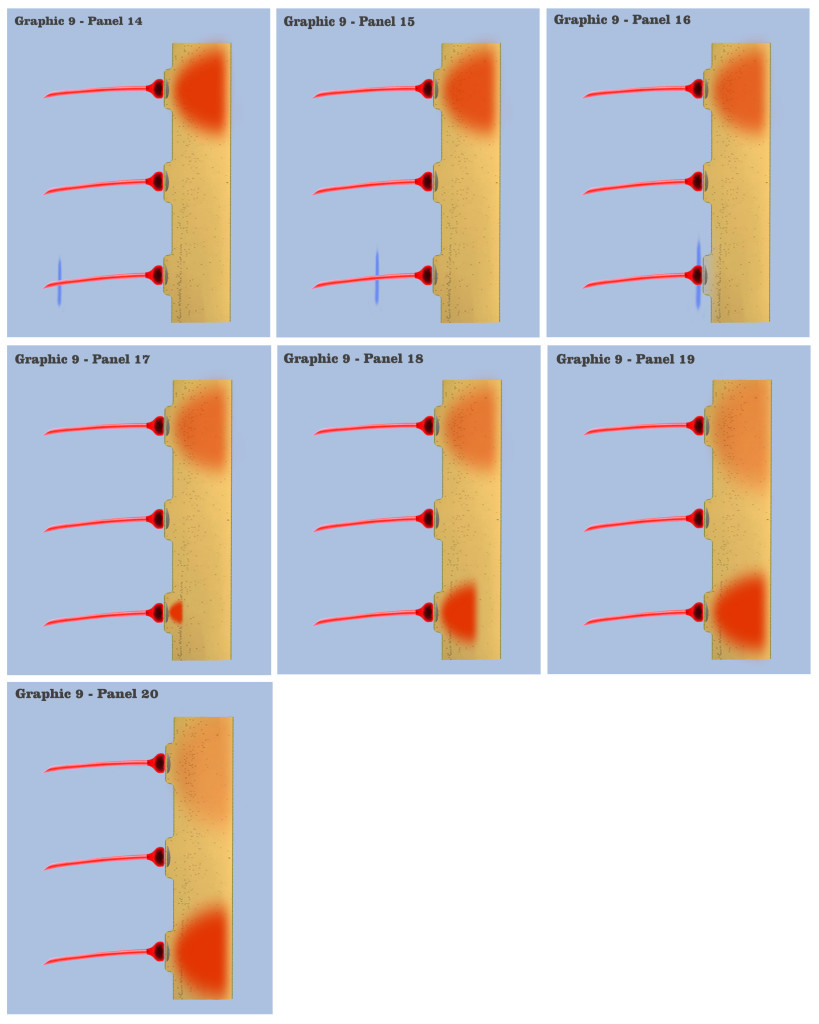
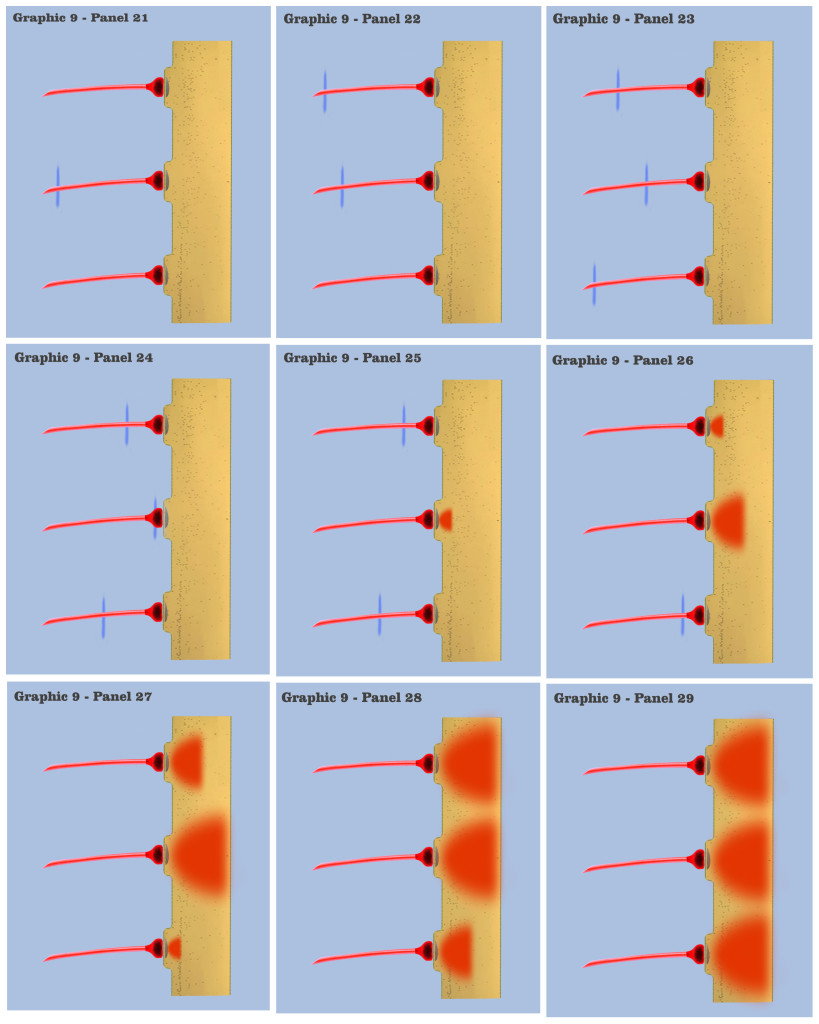
In graphic 9, we begin to visualize the collective nature of dendritic response to spatially localized multiple effective presynaptic axons. Panels 1 through 7 of graphic 9 depict the time course of an axonic action potential at the middle axon, just as we saw in the visualization for the individual charge depolarization, which is followed by an axonic action potential from the upper axon in panels 8 through 13, and finally in panels 14 through 20 we see the time course of a third axonic action potential at the lower axon, after which it should be noted that because of the electrotonic charge dissipation, even action potentials at spatially local synapses cannot accumulate their dendritic charge if the effective impulses do not arrive within a certain time window, a period the dialog is calling the charge latency, which is roughly comparable to the common neuro-physical property called the membrane time constant.
And in the final panels of graphic 9, the visualization depicts an actual charge accumulation that could lead to a charge threshold, a charge accumulation that occurs due to the inability of individual charge depolarizations to spread and dissipate, their charge potentials held in check by neighboring charge potentials.
The dialog wants to point out to the bottom-up engineer that, although somewhat simplified, this visualization depicts the temporal response for all of the neural elements we have encountered so far in the neural array of our primordial motile metazoan, a temporal response framed by the charge latency of electrotonic dissipation. Here, charge accumulation can occur in these neural elements only if the signaling at their dendrites coincides within the time period of the charge latency. And the dendritic threshold cannot be achieved without a certain measure of charge accumulation in the dendrites of any neuron.
So the bottom-up engineer should at this point see that what is needed for the next evolution in neuro-phenotype is a mechanism to bridge this charge latency time window, a mechanism which the dialog has labeled persistence.
When introducing the depiction of an individual depolarization, this discussion defined all axonic action potential impulses as temporally dimensionless, and the discussion went on to say that the ionotropic depolarization of the dendritic synapse represents the basic expression of temporal dimensionality in neurophysiological signaling. Now certainly, the persistence that the dialog has been alluding to would constitute yet another dimensionality to the temporal response in signaling, but what neurophysiological form would this next dimensionality take?
In extant mammalian species today, there are a number of postsynaptic mechanisms, all with varying temporal properties, and indeed they are traditionally classed into two distinct categories, categories that are dichotomized by the time scale in which they demonstrate their behavior. The two classes of receptor mechanisms, called ionotropic receptors and metabotropic receptors, have varying properties but are chiefly characterized by the time scale of these properties.
Ionotropic receptors, which are the evolutionary latest act of the primordial ionotropic depolarizations depicted in graphics 8 and 9, are considered to be fast-acting but with a relatively short charge latency. Metabotropic receptors, which are slower acting, requiring sometimes complex molecular intermediaries, can demonstrate a longer term response to synaptic activity than ionotropic receptors, a property that appears to be Natures’ solution for the need for persistence in synaptic signaling.
Although there are numerous metabotropic receptors, the dialog would like to focus on a particular metabotropic mechanism to use as an introduction for the new neurophysiological process that Nature would develop for our evolving primordial motile metazoan, the process which evolutionary selective pressures were demanding at the end of discussion 9f.
The metabotropic mechanism that will be focused on is centered on the Locus Coeruleus, a relatively small neural aggregate buried deep in the brainstem of extant mammalian species, a location that hints at its ancient, even primordial evolutionary origins.
As the principle site for the chemical synthesis of the neurotransmitter norepinephrine in the brain, the locus coeruleus is a nucleus that is part of what is referred to as the Reticular Activation System, a system responsible for regulating wakefulness and mediating transitions in the state of attention. It is the quintessential tonic assembly, as it literally maintains the “tone” of overall brain activity on a circadian basis, and just as importantly, as environmentally salient events are sensed, it conditions certain areas of the brain to allow heightened states of attention to these events.
This nucleus and its norepinephrine neurotransmitters have been singled out for discussion because of a number of reasons, but there are two notable aspects which merit highlighting. The nucleus itself is relatively small, with only about 50,000 neural elements comprising its output stage, but the bilateral projections of the locus coeruleus reach a widespread and extensive volume of targets throughout the brain, including much of the sensory areas of the cortex, the cerebellum and the brainstem, all except, curiously, the basal ganglia, a neural assembly for which the dialog has hinted plays a large part in the release of phylogenetically developed motoric sequences.
The second notable aspect which merits mention regards the fashion in which the neural norepinephrine afferents innervate the sensory areas of the mammalian cerebral cortex. Unlike the numerous afferents from the thalamus which innervate the many areas of the cortex primarily with projections to layer 4 of the cortex, the projections from the locus coeruleus to its various areas have branching axons that terminate in all six layers of the cortex. This aspect is significant in that it says something about the nature of the signaling from the locus coeruleus.
With only about 50,000 neural elements projecting to perhaps billions of cells throughout the brain, this signaling surely would not be characterized as the sending of specific “information” to all of these brain areas. So what is it they are communicating?
The dialog would like the bottom-up engineer to characterize this signaling from the locus coeruleus to the diverse parts of the brain to be not so much communicating to its afferent neurons, but the conditioning or biasing of these afferent neurons, a unique form of neural signaling which is characteristic not in the form of its effective axonic action potential impulses, but in the affective target of its dendritic synapses.
Mammalian attention processes are meant to focus the sensory modalities on filtered subsets of salient sensation, but there are situations where the mammalian organism necessarily must relax these attentive filters, and place its entire sensory apparatus “on guard” so to say, in order to be receptive to its entire spectrum of environmental signaling, allowing a response to sensations or signaling events previously filtered out. This “biasing” of the organisms’ neural system must be accomplished by a mechanism that is separate from the organisms’ sensory system itself. It is the conditioning signaling from the locus coeruleus that is performing in large part this separate biasing mechanism.
And since this “priming signaling” must be separate from the very sensory signaling that it is conditioning, we would expect to see Nature evolve separate mechanisms in the dendrites of neurons to institute this segregation.
Within the dendrites of extant mammalian neurons are certain intracellular organelles, called smooth endoplasmic reticulum (SER), which serve many subcellular functions. Indeed, these organelles are part of a “membrane within a membrane”, formed by a specialization inside the neural membrane that spans the neuron from dendrite to soma to axonic projection. Throughout the neuron, the endoplasmic reticulum acts as a lipid and protein transport and delivery system, and within the dendrites of neurons, the SER serves another important function, as a regulating store of calcium ions, ions which play a dual role in dendrites as charge carriers and intracellular messengers.
The storage and release of these calcium ions from the SER to within the intracellular dendritic medium can have a profound modulating effect on the behavior of extracellular ionotropic signaling to a dendrite. So it should be evident to the bottom-up engineer that Nature has crafted a way to control the storage and release of these modulating ions either intracellularly through intracellular messengers, or extracellularly, through the metabotropic mechanisms that are being introduced.
Returning to the locus coeruleus, the effective axons of the locus coeruleus nucleus terminate their many synapses with a metabotropic neurotransmitter, norepinephrine, which does not produce ionotropic depolarizations as we visualized in graphics 8 and 9. The neurotransmitters at these synapses set in motion a more complex chain of molecular events, which implements a subcellular “biasing” that the locus coeruleus system has evolved to establish. And one of those biasing processes is the conditioning of the endoplasmic reticulum in these target neurons to vary their storage and release of calcium ions.
It is the cycle of intracellular calcium ion sequestration that modulates a neurons’ ionotropic signaling, caused in this case by the metabotropic response to the norepinephrine neurotransmitter, which finally returns the dialog back to the discussion on primordial persistence that has taken such a circuitous route to get to. Now, the neural arrays of our primordial motile metazoans do not possess any of the contemporary mammalian functions that were just discussed, such as attention mechanisms or sensory filters, but it will be instructive to the bottom-up engineer to see how the metabotropic conditioning apparatus of the present day locus coeruleus system, and many other mammalian metabotropic processes, might have evolved from the primordial persistence mechanisms that we began this discussion with. So let us “rewind the evolution” of the locus coeruleus back to the point in our imagination scenario of the primordial metazoan when the adaptation of this neurological functionality may have begun.
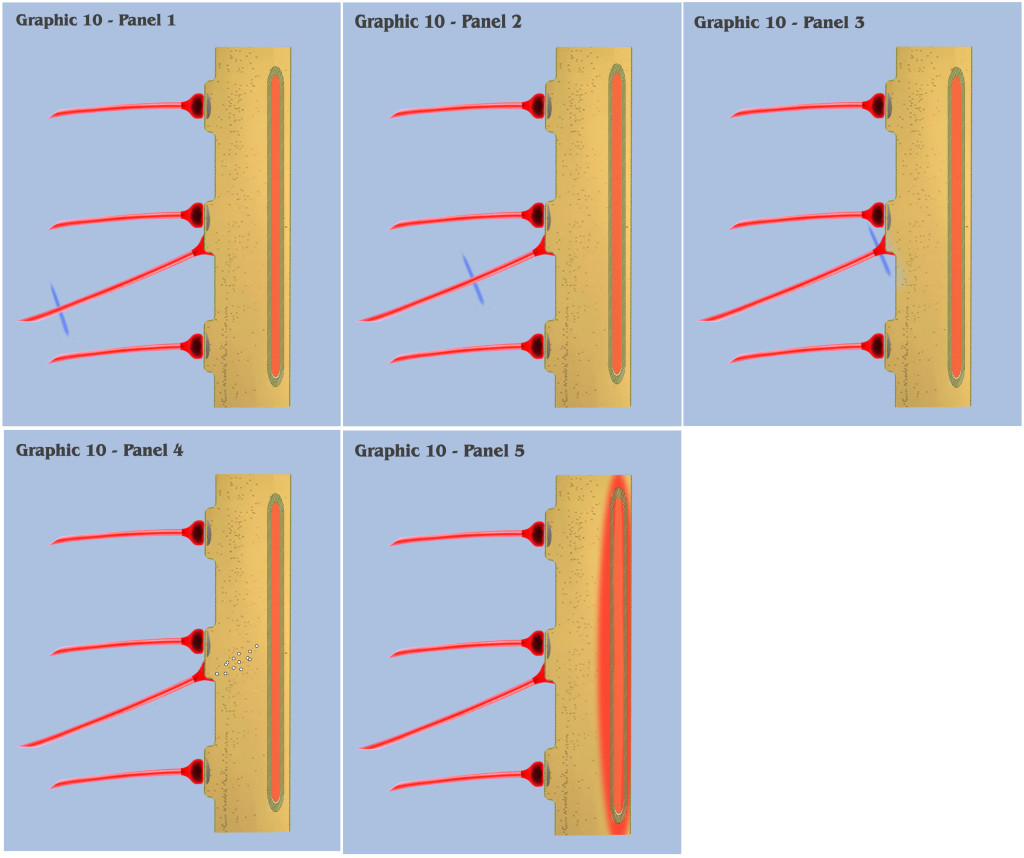
With graphic 10, we return to the visualization of primordial ionotropic depolarization and charge accumulation, but with the addition of a new element, the newly evolved primordial metabotropic neural axon. (Although not depicted, the bottom-up engineer should view this new effective axon as projecting from a newly evolved metabotropic neuron, the primordial counterpart to the locus coeruleus neuron we see in extant mammals today. However, the dialog will not address the phylogenesis of primordial metabotropic neurons until the discussion on the third fundamental precept.)
In panel 1 of graphic 10, the approaching action potential impulse is depicted as traveling along the metabotropic axon, where the bottom-up engineer will notice the addition of a second new element to the visualization. Within the dendrite is depicted the primordial endoplasmic reticulum, an organelle which has evolved to express some, but not all, of the dendritic modulating properties that we see in extant mammalian neurons today.
The properties that our primordial endoplasmic reticulum so far have evolved is limited to the ability to collect and store excess calcium ions allowed into the dendritic membrane by past ionotropic depolarizations, with the secondary property of a mechanism to release its stored ions back into the dendritic medium. The mechanism that controls the storage of ions is as yet unspecified and not relevant to this discussion, however, it is the mechanism by which the endoplasmic reticulum releases calcium ions that is the subject of this graphic.
In panel 2 of graphic 10, we see the metabotropic action potential traveling further along the projecting axon, and in panel 3 we see the action potential as it arrives at the presynaptic bouton. At this point, all of the presynaptic mechanisms which occur for ionotropic signaling are identical here, with the exception that metabotropic neurotransmitters are released by the presynaptic bouton, as opposed to the ionotropic neurotransmitters previously discussed.
It is at panel 4 of graphic 10 that we witness the first neurophysiological variation in metabotropic signaling over the previously visualized ionotropic signaling. Instead of forming a charge potential by “opening the gate” to extracellular charge ions, the postsynaptic metabotropic receptor in our new neuron cytophysiology sets into motion a chain of molecular reactions, which contemporary neuroscientists have labeled “second messenger” reactions.
And in panel 5, we see that the second messenger reactions have reached the primordial endoplasmic reticulum, where it signals the organelle to release a certain concentration of calcium ions from its intracellular store into the dendritic medium.
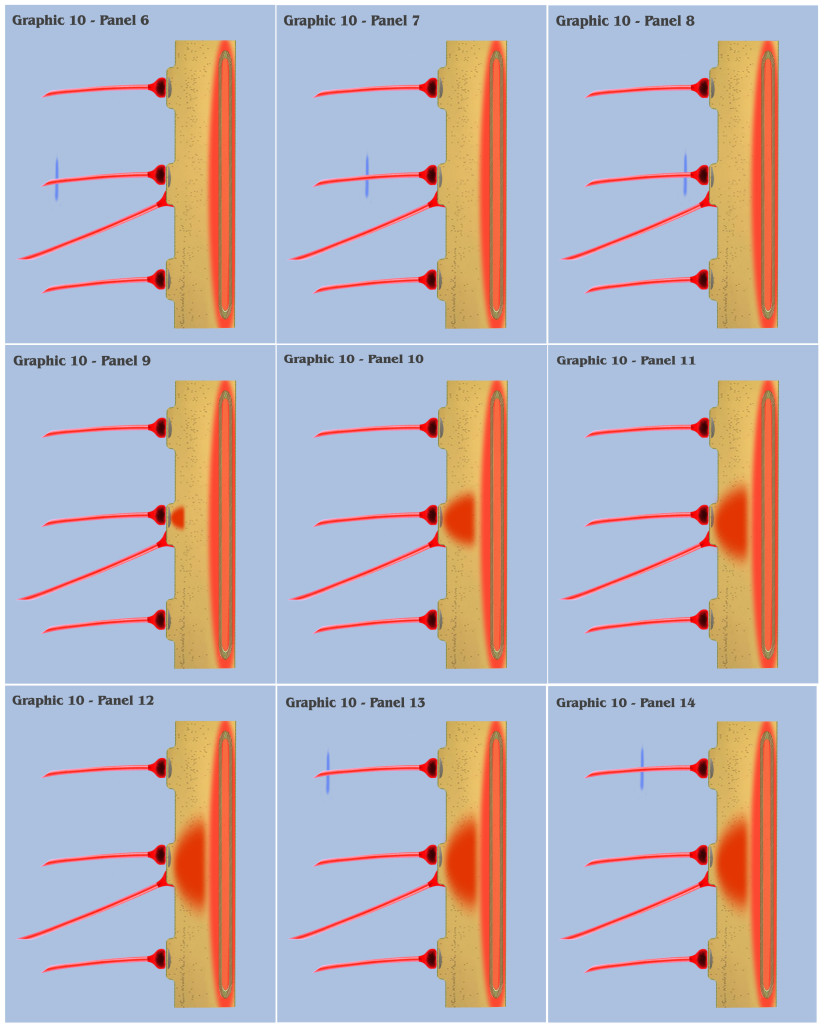
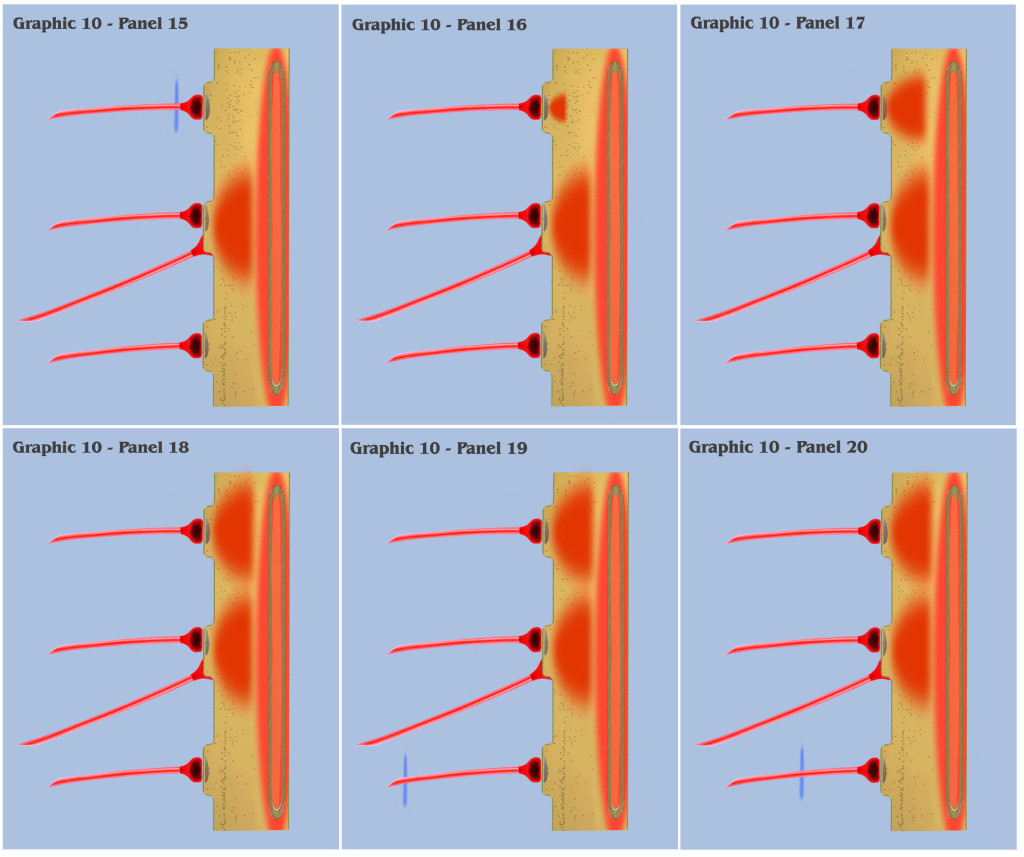
At this point, the visualization would like to contrast panels 6 through 25 of graphic 10 with panels 1 through 20 of graphic 9. In panels 1 through 20 of graphic 9, the visualization depicted the progression as three spatially local axons transmitted three temporally disjoint action potential impulses to our primordial affective dendrite, and because of the electrotonic charge dissipation, the three disjoint charge potentials produced in the dendrite could not accumulate to trigger a charge threshold. Panels 6 through 25 of graphic 10 depict an identical time progression as in panels 1 through 20 of graphic 9, but the bottom-up engineer should notice that the individual charge depolarizations of the three synapses in graphic 10 are following a different time course than they did in graphic 9. Because of the intracellular charge field created by the metabotropically activated endoplasmic reticulum, the charge fields of the three temporally disjoint ionotropic depolarizations in graphic 10 do not have a “charge vacuum” to disperse into, and thus do not dissipate until a much longer time period.
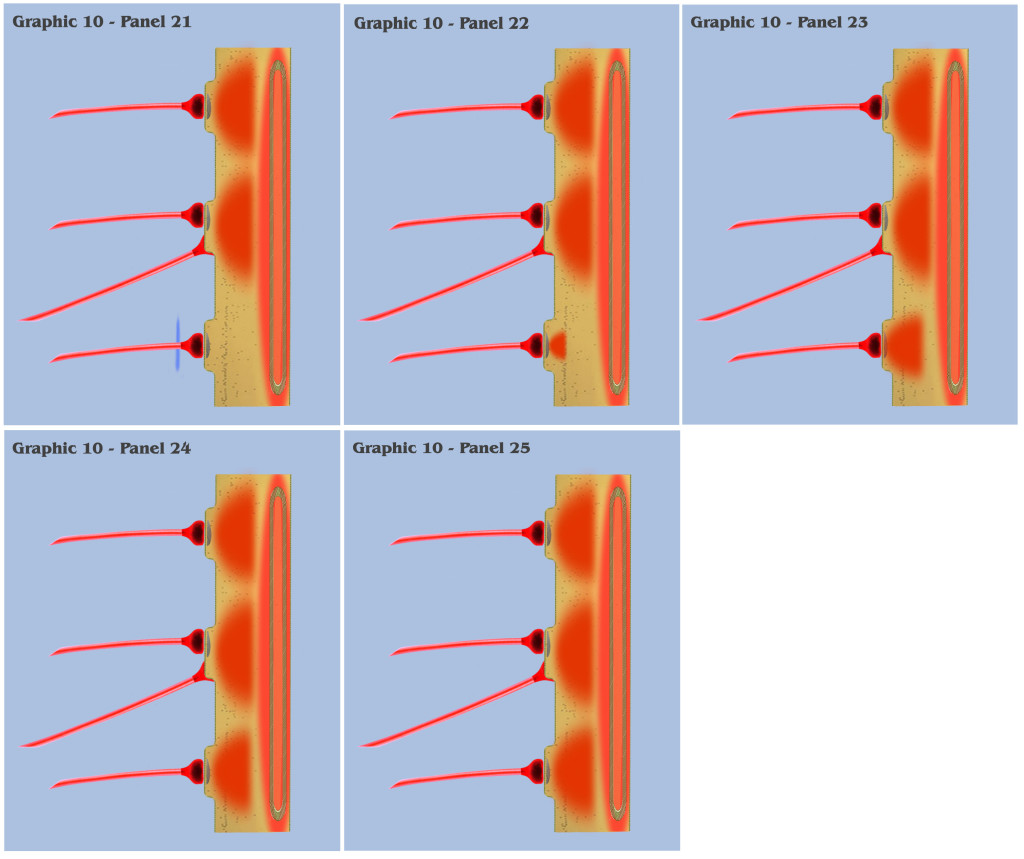
This longer charge latency allows the charge fields to accumulate, just as if they were temporally synchronized, which subsequently triggers a synthetic dendritic threshold.
In conclusion of the visualization that began with graphic 6, the depiction that progressed through to the panels in graphic 10 illustrate the neurophysiological form that Nature might have developed to implement the needed mechanism of persistence in sensory signaling.
And because this new mechanism must leave intact the functionality of the basic sensory apparatus based on ionotropic signaling, whose neural elements that collect instant information from the environment must function without modification by any prior sensation or signaling, this new mechanism adapted a form which performed as a separate construction that conditioned the existing basic circuitry to behave in a persistent manner only during controlled periods by a phylogenetic mechanism (the future attention mechanism), that would in time evolve into the multi-variant modes of metabotropic neural signaling that we find in extant mammalian species today.
The selective pressures to develop a mechanism of persistence came about from the dilemma of the dangling direction vector, in which our evolving motile metazoan, after having evolved photonic sensation, had produced the ability to apprehend shadow direction, but with no emotive sense to enable its exploitation. This emotive component to the shadow direction signaling had to be somehow abstracted, or created from a synthetic apprehension of the available sensory signaling. This synthetic apprehension, which is considered to be elementary abstraction, shall be the next step which forms the definition of state in the organism.
So, at this point, there are two lessons that the dialog needs to present to the bottom-up engineer. For the first lesson, the dialog needs to return to the imagination scenario of our primordial metazoan, and show how the newly evolved mechanism of persistence can resolve the dilemma of the dangling direction vector, to further the evolution of the metazoans’ emotive complex into a cognitive complex, a new complex that will be prompted by the second lesson.
That second lesson will be to show how persistence will form the basic substrate for our evolving metazoan to climb the next rung in the ladder of dimensional resolution, a rung which can be characterized as a resolution of the temporal dimension in experience by this newly evolving mechanism of primordial metabotropic transmission.
GO TO NEXT DISCUSSION
GO TO TOP OF DISCUSSION
Copyright © 2019 All rights reserved